PHARMACEUTICAL DEVELOPMENT
I. INTRODUCTION
II. PHARMACEUTICAL DEVELOPMENT
A. Components of the Drug Product
1. Drug Substance
2. Excipients
B. Drug Product
1. Formulation Development
2. Overages
3. Physicochemical and Biological Properties
C. Manufacturing Process Development
D. Container Closure System
E. Microbiological Attributes
F. Compatibility
III. GLOSSARY
INTRODUCTION
The Pharmaceutical Development provides an opportunity to present the knowledge gained through the application of scientific approaches and quality risk management to the development of a product and its manufacturing process. It is first produced for the original marketing application and can be updated to support new knowledge gained over the lifecycle of a product.
The Pharmaceutical Development is intended to provide a comprehensive understanding of the product and manufacturing process for reviewers and inspectors and indicates areas where the demonstration of greater understanding of pharmaceutical and manufacturing sciences can create a basis for flexible regulatory approaches.
PHARMACEUTICAL DEVELOPMENT:
Pharmaceutical development is to design a quality product and its manufacturing process to consistently deliver the intended performance of the product. The information and knowledge gained from pharmaceutical development studies and manufacturing experience provide scientific understanding to support the establishment of the design space, specifications, and manufacturing controls and information from pharmaceutical development studies can be a basis for quality risk management.
It is important to recognize that quality cannot be tested into products, i.e., quality should be built in by design. Changes in formulation and manufacturing processes during development and lifecycle management should be looked upon as opportunities to gain additional knowledge and further support establishment of the design space. Similarly, inclusion of relevant knowledge gained from experiments giving unexpected results can also be useful.
Design space is proposed by the applicant and is subject to regulatory assessment and approval. Working within the design space is not considered as a change. Movement out of the design space is considered to be a change and would normally initiate a regulatory postapproval change process.
To establishes the type of dosage form and the formulation proposed are suitable for the intended use and include sufficient information to provide an understanding of the development of the drug product and its manufacturing process.
At a minimum, those aspects of drug substances, excipients, container closure systems, and manufacturing processes that are critical to product quality should be determined and control strategies justified. Critical formulation attributes and process parameters are identified through an assessment to which their variation can have impact on the quality of the drug product.
The applicant can choose to conduct pharmaceutical development studies that can lead to an enhanced knowledge of product performance over a wider range of material attributes, processing options and process parameters.
This scientific understanding facilitates establishment of an expanded design space. In these situations, opportunities exist to develop more flexible regulatory approaches, for example, to facilitate:
- Risk-based regulatory decisions (reviews and inspections)
- Manufacturing process improvements, within the approved design space described in the dossier, without further regulatory review
- Reduction of postapproval submissions
- Real-time quality control, leading to a reduction of end-product release testing
To realize this flexibility, the applicant should demonstrate an enhanced knowledge of product performance over a range of material attributes, manufacturing process options and process parameters.This knowledge can be gained by application of, for example, formal experimental designs, process analytical technology (PAT), and/or prior knowledge.
The design and conduct of pharmaceutical development studies should be consistent with their intended scientific purpose. It should be recognized that the level of knowledge gained, and not the volume of data, provides the basis for science-based submissions and their regulatory evaluation.
A. Components of the Drug Product
1. Drug Substance
The physicochemical and biological properties of the drug substance that can influence the performance of the drug product and its manufacturability, or were specifically designed into the drug substance (e.g., solid state properties), should be identified and evaluated.
Examples of physicochemical and biological properties that should be examined, include:
Solubility,
Water content,
Particle size,
Crystal properties,
Biological activity, and
Permeability.
These properties could be interrelated and should be considered in combination.
To evaluate the potential effect of drug substance physicochemical properties on the performance of the drug product, studies on drug product might be warranted.
For example, ICH Q6A Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances describes some of the circumstances in which drug product studies are recommended (e.g., Decision Trees #3 and #4 (Part 2)).
This approach applies equally for ICH Q6B Specifications: Test Procedures and Acceptance Criteria for Biotechnology/Biological Products.
The knowledge gained from the studies investigating the potential effect of drug substance properties on drug product performance can be used, to justify elements of the drug substance specification.
The compatibility of the drug substance with excipients should be evaluated.
For products that contain more than one drug substance, the compatibility of the drug substances with each other should also be evaluated.
2. Excipients:
The excipients,their concentration and the characteristics that can influence the drug product performance (e.g., stability, bioavailability) or manufacturability should be consider relative to the respective function of each excipient.
All substances used in the manufacture of the drug product, whether they appear in the finished product or not (e.g., processing aids).
Compatibility of excipients with other excipients, where relevant (for example, combination of preservatives in a dual preservative system), should be established.
The ability of excipients (e.g., antioxidants, penetration enhancers, disintegrants, release controlling agents) to provide their intended functionality and to perform throughout the intended drug product shelf life should also be demonstrated.
The excipient performance can be used to justify the choice and quality attributes of the excipient and to support the justification of the drug product specification.
Information to support the safety of excipients should be cross-referenced.
B. Drug Product
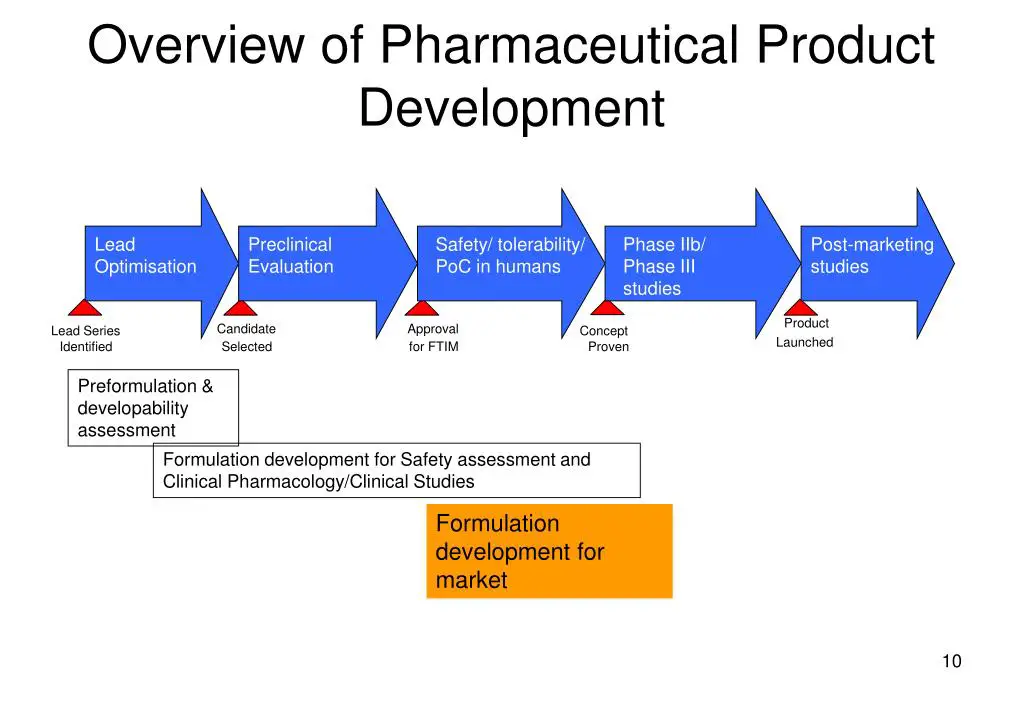
1. Formulation Development
A summary should be provided describing the development of the formulation, including identification of those attributes that are critical to the quality of the drug product, taking into consideration intended usage and route of administration.
The formal experimental designs can be useful in identifying critical or interacting variables that might be important to ensure the quality of the drug product.
The summary should highlight the evolution of the formulation design from initial concept up to the final design and also take into consideration the choice of drug product components (e.g., the properties of the drug substance, excipients, container closure system, any relevant dosing device), the manufacturing process and knowledge gained from the development of similar drug products.
Any excipient ranges included in the batch formula should be justified in the Pharmaceutical Development section of the application, this justification can often be based on the experience gained during development or manufacture.
A summary of formulations used in clinical safety and efficacy and in any relevant bioavailability or bioequivalence studies should be provided.
Any changes between the proposed commercial formulation and those formulations used in pivotal clinical batches and primary stability batches should be clearly mentioned and the rationale for the changes provided.
Information from comparative in vitro studies (e.g., dissolution) or comparative in vivo studies (e.g., bioequivalence) that links clinical formulations to the proposed commercial formulation should be summarized and a cross-reference to the studies (with study numbers) should be provided.
Where attempts have been made to establish an in vitro/in vivo correlation, the results of those studies and a cross-reference to the studies (with study numbers) should be provided in the Pharmaceutical Development.
A successful correlation can assist in the selection of appropriate dissolution acceptance criteria and can potentially reduce the need for further bioequivalence studies following changes to the product or its manufacturing process.
Any special design features of the drug product (e.g., tablet score line, overfill, anticounterfeiting measure as it affects the drug product) should be identified and a rationale provided for their use.
2. Overages
Use of an overage of a drug substance to compensate for degradation during manufacture or a product’s shelf life, or to extend shelf life, is discouraged.
Any overages in the manufacture of the drug product, whether they appear in the final formulated product or not, should be justified considering the safety and efficacy of the product.
Information should be provided on the
(1) amount of overage,
(2) reason for the overage (e.g., to compensate for expected and documented manufacturing losses), and
(3) justification for the amount of overage.
The overage should be included in the amount of drug substance listed in the batch formula.
3. Physicochemical and Biological Properties
The physicochemical and biological properties relevant to the safety, performance, or manufacturability of the drug product should be identified and discussed. This includes the physiological implications of drug substance and formulation attributes.
For example, the development of a test for respirable fraction of an inhaled product.
Similarly, information supporting the selection of dissolution vs. disintegration testing (or other means to ensure drug release) and the development and suitability of the chosen test could be provided in this section.
See also ICH Q6A Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances, Decision Tree #4 (Part 3) and Decision Tree #7 (Part 1) or ICH Q6B Specifications: Test Procedures and Acceptance Criteria for Biotechnology/Biological Products.
The discussion should cross-reference any relevant stability data.
C. Manufacturing Process Development
The control and any improvement of the manufacturing process (i.e., intended for commercial production batches) should be explained. It is important to consider the critical formulation attributes, together with the available manufacturing process options, in order to address the selection of the manufacturing process and confirm the appropriateness of the components.
Appropriateness of the equipment used for the intended products should be discussed. Process development studies should provide the basis for process improvement, process validation, continuous process verification (where applicable), and any process control requirements.
Where appropriate, such studies should address microbiological as well as physical and chemical attributes. The knowledge gained from process development studies can be used to justify the drug product specification.
The manufacturing process development program or process improvement program should identify any critical process parameters that should be monitored or controlled (e.g., granulation end point) to ensure that the product is of the desired quality.
For those products intended to be sterile, an appropriate method of sterilization for the drug product and primary packaging material should be chosen and the choice justified.
Significant differences between the manufacturing processes used to produce batches for pivotal clinical trials (safety, efficacy, bioavailability, bioequivalence) or primary stability studies and the process should be discussed.
The discussion should summarize the influence of the differences on the performance, manufacturability, and quality of the product. The information should be presented in a way that facilitates comparison of the processes and the corresponding batch analyses information .
The information should include, for example,
(1) the identity (e.g., batch number) and use of the batches produced (e.g., bioequivalence study batch number),
(2) the manufacturing site,
(3) the batch size, and
(4) any significant equipment differences (e.g., different design, operating principle, size).
In order to provide flexibility for future process improvement, when describing the development of the manufacturing process, it is useful to describe measurement systems that allow monitoring of critical attributes or process end-points.
Collection of process monitoring data during the development of the manufacturing process can provide useful information to enhance process understanding. The process control strategies that provide process adjustment capabilities to ensure control of all critical attributes should be described.
An assessment of the ability of the process to reliably produce a product of the intended quality (e.g., the performance of the manufacturing process under different operating conditions, at different scales, or with different equipment) can be provided.
An understanding of process robustness can be useful in risk assessment and risk reduction (see ICH Q9 Quality Risk Management glossary for definition) and to support future manufacturing and process improvement, especially in conjunction with the use of risk management tools (see ICH Q9 Quality Risk Management).
D. Container Closure System
The choice and rationale for selection of the container closure system for the commercial product should be discussed. Consideration should be given to the intended use of the drug product and the suitability of the container closure system for storage and transportation (shipping), including the storage and shipping container for bulk drug product, where appropriate.
The choice of materials for primary packaging should be justified. The discussion should describe studies performed to demonstrate the integrity of the container and closure. A possible interaction between product and container or label should be considered.
The choice of primary packaging materials should consider, e.g., choice of materials, protection from moisture and light, compatibility of the materials of construction with the dosage form (including sorption to container and leaching), and safety of materials of construction. Justification for secondary packaging materials should be included, when relevant.
If a dosing device is used (e.g., dropper pipette, pen injection device, dry powder inhaler), it is important to demonstrate that a reproducible and accurate dose of the product is delivered under testing conditions that, as far as possible, simulate the use of the product.
E. Microbiological Attributes
The microbiological attributes of the drug product should be discussed. The discussion should include, for example:
The rationale for performing or not performing microbial limits testing for non sterile drug products (e.g., Decision Tree #8 in ICH Q6A Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances and ICH Q6B Specifications: Test Procedures and Acceptance Criteria for Biotechnology/Biological Products)
• The selection and effectiveness of preservative systems in products containing antimicrobial preservative or the antimicrobial effectiveness of products that are inherently antimicrobial.
• For sterile products, the integrity of the container closure system as it relates to preventing microbial contamination.
Chemical testing for preservative content is the attribute normally included in the drug product specification, antimicrobial preservative effectiveness should be demonstrated during development. The lowest specified concentration of antimicrobial preservative should be demonstrated to be effective in controlling microorganisms by using an antimicrobial preservative effectiveness test.
The concentration used should be justified in terms of efficacy and safety, such that the minimum concentration of preservative that gives the required level of efficacy throughout the intended shelf life of the product is used. Where relevant, microbial challenge testing under testing conditions that, as far as possible, simulate patient use should be performed during development and documented in this section.
F. Compatibility
The compatibility of the drug product with reconstitution diluents (e.g., precipitation, stability) should be addressed to provide appropriate and supportive information for the labeling.
This information should cover the recommended in-use shelf life, at the recommended storage temperature and at the likely extremes of concentration. Similarly, admixture or dilution of products prior to administration (e.g., product added to large volume infusion containers) should be addressed.
III. GLOSSARY
Continuous Process Verification: An alternative approach to process validation in which manufacturing process performance is continuously monitored and evaluated.
Design Space: The multidimensional combination and interaction of input variables (e.g., material attributes) and process parameters that have been demonstrated to provide assurance of quality. Working within the design space is not considered as a change. Movement out of the design space is considered to be a change and would normally initiate a regulatory postapproval change process. Design space is proposed by the applicant and is subject to regulatory assessment and approval.
Formal Experimental Design: A structured, organized method for determining the relationship between factors affecting a process and the output of that process. Also known as “Design of Experiments.”
Lifecycle: All phases in the life of a product from the initial development through marketing until the product’s discontinuation.
Process Analytical Technology (PAT): A system for designing, analyzing, and controlling manufacturing through timely measurements (i.e., during processing) of critical quality and performance attributes of raw and in-process materials and processes with the goal of ensuring final product quality.
Process Robustness: Ability of a process to tolerate variability of materials and changes of the process and equipment without negative impact on quality.
Quality: The suitability of either a drug substance or drug product for its intended use. This term includes such attributes as the identity, strength, and purity (from ICH Q6A Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances).
The use of the word should in Agency guidance’s means that something is suggested or recommended, but not required.
For Details Information, please refer Guidance for Industry -Q8(R2) Pharmaceutical Development.
Reference: Guidance for Industry -Q8(R2) Pharmaceutical Development.