Objective: To lay down a procedure for calibration of pH Meter. Scope: This SOP is applicable for calibration of pH Meter in quality control laboratory. Responsibility: Officer or above of QC laboratory is responsible for calibration of pH Meter Head Quality / nominee of QC department is responsible for reviewing …
Read More »Sampling and testing of Sterile surgical Gloves
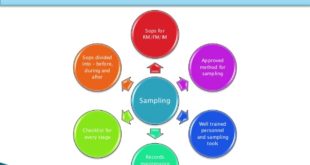
Objective: To lay down the procedure for Sampling and testing of sterile surgical gloves. Scope: This SOP is applicable for sampling and testing of sterile surgical gloves. Responsibility: Personnel (Microbiology-QC) shall be responsible for sampling and testing of sterile surgical gloves. Executive/ Officer shall be responsible for ensuring the compliance …
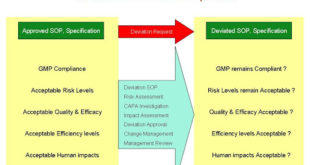
Read More »Deviation Handling and Quality Risk Management
PPT on prevent the spread of COVID 19 infection at the workplace
Investigating Out-of-Specification
Investigating Out-of-Specification IDENTIFYING AND ASSESSING OOS TEST RESULTS — PHASE I: LABORATORY INVESTIGATION PHASE II: FULL-SCALE OOS INVESTIGATION
Read More »PROCEDURE FOR CALIBRATION OF REFRIGERATOR
OBJECTIVE:To describe the calibration procedure for distribution of temperature within the chamber of Refrigerator. SCOPE: his SOP shall be applicable for Calibration of Refrigerator at Pharmaceutical Industries. RESPONSIBILITY : Quality Control Executive/Officer. ACCOUNTABILITY Head Quality Control PROCEDURE : Operate the refrigerator as per Standard …
Read More »USFDA GUIDELINE FOR DRUG QUALITY SAMPLING AND TESTING (DQST)- HUMAN DRUGS
USFDA GUIDELINE FOR DRUG QUALITY SAMPLING AND TESTING (DQST)- HUMAN DRUGS
Read More »List of Documents required for Pharma Quality Control Departments (GMP & GLP Compliances)
List of Documents required for Quality Assurance Departments (GMP Compliances)
STABILITY STUDY MANAGEMENT
STABILITY STUDY MANAGEMENT Stability Study Management – To lay down the procedure to conduct stability studies of the drug product (dosage forms)/ Drug substances (API). This Procedure describes the procedure for assigning the stability of drug substances (Active pharma ingredients) and drug products (Packed finished dosage forms). The purpose of …
Read More »In-process control of oral drug product during manufacturing & Packing
Objective To lay down the procedure for in-process control of oral drug products during manufacturing & Packing. Scope This procedure is applicable for in-process sampling, analysis, and reporting to be carried out during manufacturing and packing of drug products at the formulation Plant. Responsibility Quality Assurance and production personnel shall …
Read More »SAMPLING OF PACKING MATERIALS
OBJECTIVE To lay down a procedure for sampling of packaging materials. SCOPE To describe the procedure for sampling of Primary packing materials i.e. Aluminium Foil, Blister Aluminium Foil, PVC Film etc. and secondary packing materials i.e. Cartons, labels, Leaflet shipper etc. RESPONSIBILITY Quality Control Executive/Officer ACCOUNTABILITY Quality Assurance Manager PROCEDURE …
Read More »PROCEDURE FOR OPERATION OF UNIT DOSAGE SAMPLER
1.0 OBJECTIVE To lay down the procedure for operation and cleaning of unit dose sampler. 2.0 SCOPE This SOP shall be applicable for IPQA area in Quality Assurance. 3.0 RESPONSIBILITY In process Quality Assurance Executive /Officer 4.0 ACCOUNTABILITY Head Quality Assurance 5.0 PROCEDURE FOR OPERATING 5.1 Check the Status …
Read More »Batch Release Statement for Pharmaceutical Product
Procedure for Microbiological Monitoring of Purified water
SOP In-process Control of Packing Lines
SOP on Sampling of Intermediates and Finished Products
SOP On In-process Control During Tablets Manufacturing
SOP on Line Clearance
SOP on QUALITY RISK MANAGEMENT (RISK ASSESSMENT)
1.0 Objective: To provide a procedure for carrying out Risk assessment, evaluation, mitigation and review of risk by employing appropriate tool of Quality Risk Management Process. 2.0 Scope: Applicable to different aspects of pharmaceutical quality like development, manufacturing, testing, distribution, inspection and submission/review processes throughout the life cycle of drug …
Read More »