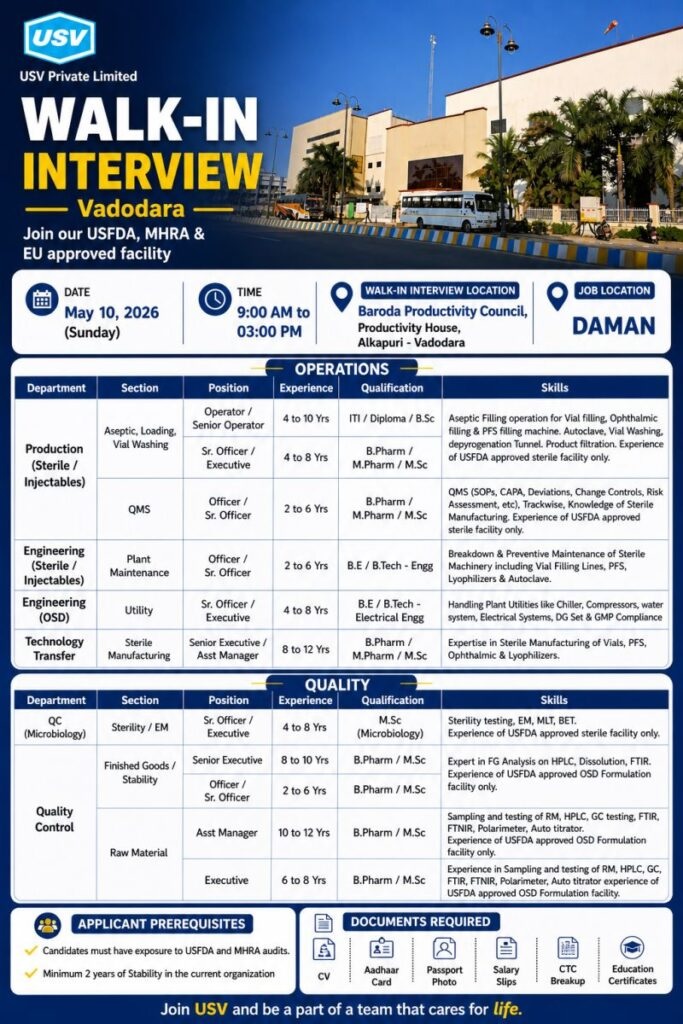
USV Private Limited is a prominent Indian multinational pharmaceutical and biotechnology company founded in 1961, specializing in diabetes and cardiovascular therapeutics. Headquartered in Mumbai, it is a market leader in Oral Anti-Diabetic (OAD) drugs and offers a diverse portfolio, including APIs, biosimilars, and injectables, alongside consumer healthcare products like Sebamed.
Join USFDA, MHRA and EU approved facility
OPERATIONS
Production (Sterile / Injectables)
Aseptic, Loading, Vial Washing QMS
Operator / Senior Operator
Experience : 4 to 10 years
Qualification : ITI / Diploma / B.Sc
Sr. Officer / Executive
Experience : 4 to 8 Yrs
Qualification : B.Pharm / M.Pharm / M.Sc
Skills : Aseptic Filling operation for Vial filling. Ophthalmic filling & PFS filling machine. Autoclave, Vial Washing, depyrogenation Tunnel. Product filtration. Experience of USFDA approved sterile facility only.
Technology Transfer
Sterile Manufacturing
Senior Executive / Asst Manager
Experience : 8 to 12 Yrs
Qualification : B.Pharm / M.Pharm / M.Sc
Skills : Expertise in Sterile Manufacturing of Vials, PFS. Ophthalmic & Lyophilizers.
QUALITY
QC (Microbiology)
Sterility / EM
Sr. Officer / Executive
Experience : 4 to 8 Yrs
Qualification : M.Sc (Microbiology)
Skills : Sterility testing, EM, MLT, BET. Experience of USFDA approved sterile facility only.
Finished Goods / Stability
Senior Executive
Experience : 8 to 10 years
BPharm / MSc
Officer / Sr.Officer
Experience : 2 to 6 years
Qualification : BPharm / MSc
Skills : Expert in FG Analysis on HPLC, Dissolution, FTIR, FTNIR, Polarimeter, Auto titrator.
Experience of USFDA approved OSD Formulation facility only.
Raw Material
Asst Manager
Experience : 10 to 12 years
Qualification : BPharm / MSc
Skills :Sampling and testing of RM, HPLC, GC testing, FTIR, FTNIR, Polarimeter, Auto titrator, Experience of USFDA approved OSD Formulation facility only.
Executive
Experience : 6 to 8 Yrs
Qualification : B.Pharm / M.Sc
Skills : Experience in Sampling and testing of RM, HPLC, GC, FTIR, FTNIR. Polarimeter, Auto titrator experience of USFDA approved OSD Formulation facility.
Jo Location : Daman
Walk in Interview
Date : 10th May 2026 (Sunday)
Time : 9 am to 3 pm
Venue : Baroda Productivity Council, Productivity House,
Alkapuri – Vadodara
APPLICANT PREREQUISITES
Candidates must have exposure to USFDA and MHRA audits.
Minimum 2 years of Stability in the current organization
Documents Required
CV, Aadhaar Card, Salary Slips, CTC Breakup, Passport photo, Education Certificates