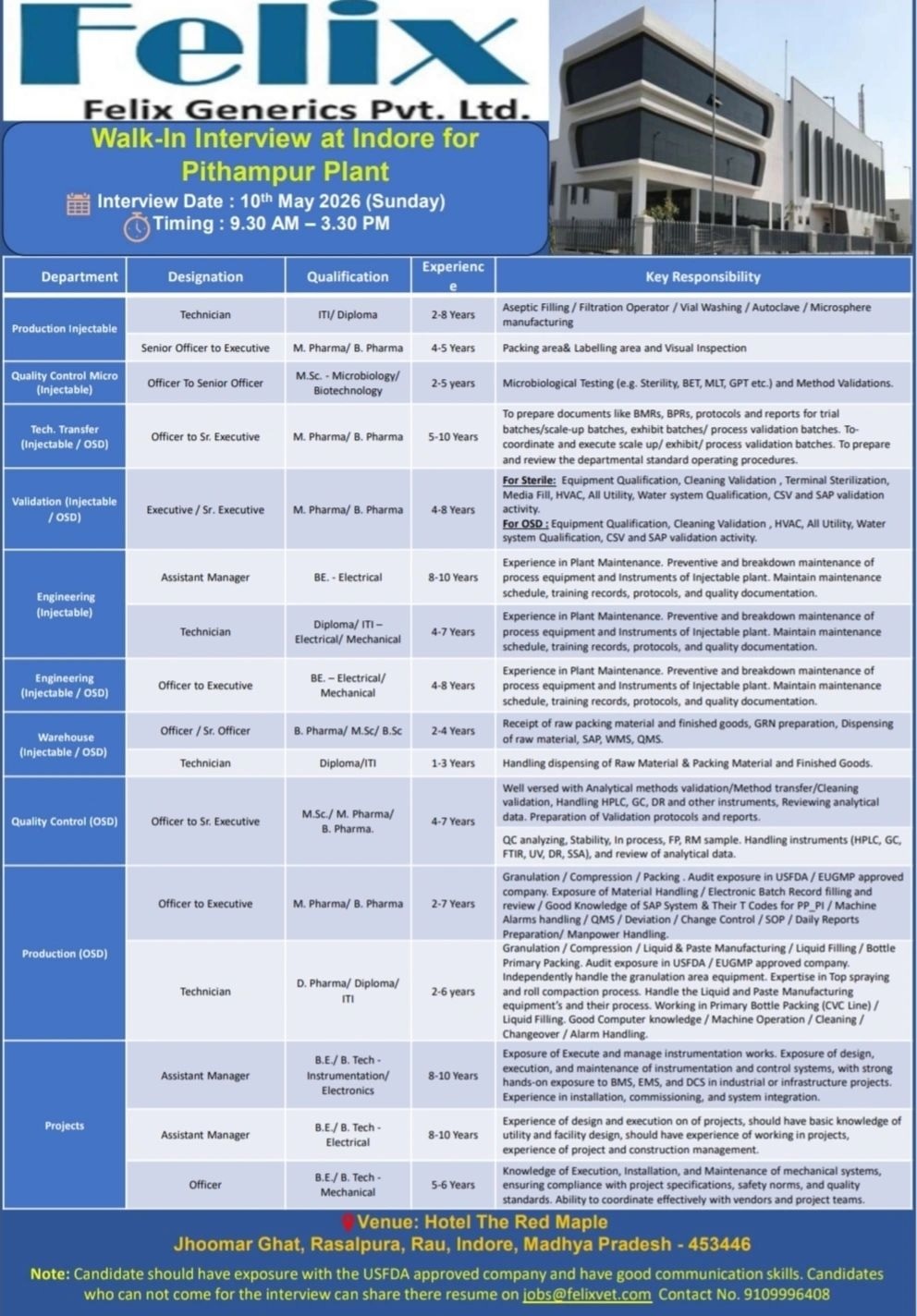
Felix Generics Pvt Ltd- Mega Walk-in Interview on 10th May 2026@ Pithampur Plant
Department: Production Injectable
Designation: Technician
Experience: 2-8 Years
Qualification: ITI/Diploma
Key skill or job description: Aseptic Filling / Filtration Operator / Vial Washing / Autoclave / Microsphere manufacturing
2. Department: Production Injectable
Designation: Senior Officer to Executive
Experience: 4-5 Years
Qualification: M. Pharma/ B. Pharma
Key skill or job description: Packing area & Labelling area and Visual Inspection
3. Department: Quality Control Micro (Injectable)
Designation: Officer To Senior Officer
Experience: 2-5 years
Qualification: M.Sc. – Microbiology/ Biotechnology
Key skill or job description: Microbiological Testing (e.g. Sterility, BET, MLT, GPT etc.) and Method Validations.
4. Department: Tech. Transfer (Injectable / OSD)
Designation: Officer to Sr. Executive
Experience: 5-10 Years
Qualification: M. Pharma/ B. Pharma
Key skill or job description: To prepare documents like BMRs, BPRs, protocols and reports for trial batches/scale-up batches, exhibit batches/ process validation batches. To coordinate and execute scale up/ exhibit/ process validation batches. To prepare and review the departmental standard operating procedures.
5. Department: Validation (Injectable / OSD)
Designation: Executive / Sr. Executive
Experience: 4-8 Years
Qualification: M. Pharma/ B. Pharma
Key skill or job description: For Sterile: Equipment Qualification, Cleaning Validation, Terminal Sterilization, Media Fill, HVAC, All Utility, Water system Qualification, CSV and SAP validation activity. For OSD: Equipment Qualification, Cleaning Validation, HVAC, All Utility, Water system Qualification, CSV and SAP validation activity.
6. Department: Engineering (Injectable)
Designation: Assistant Manager
Experience: 8-10 Years
Qualification: BE – Electrical
Key skill or job description: Experience in Plant Maintenance. Preventive and breakdown maintenance of process equipment and instruments of Injectable plant. Maintain maintenance schedule, training records, protocols, and quality documentation.
7. Department: Engineering (Injectable)
Designation: Technician Experience: 4-7 Years
Qualification: Diploma/ ITI – Electrical/ Mechanical
Key skill or job description: Experience in Plant Maintenance. Preventive and breakdown maintenance of process equipment and instruments of Injectable plant. Maintain maintenance schedule, training records, protocols, and quality documentation.
8. Department: Engineering (Injectable / OSD)
Designation: Officer to Executive
Experience: 4-8 Years
Qualification: BE – Electrical/ Mechanical
Key skill or job description: Experience in Plant Maintenance. Preventive and breakdown maintenance of process equipment and instruments of Injectable plant. Maintain maintenance schedule, training records, protocols, and quality documentation.
9. Department: Warehouse (Injectable / OSD)
Designation: Officer / Sr. Officer
Experience: 2-4 Years
Qualification: B. Pharma/ M.Sc/ B.Sc
Key skill or job description: Receipt of raw packing material and finished goods, GRN preparation, Dispensing of raw material, SAP, WMS, QMS.
10. Department: Warehouse (Injectable / OSD)
Designation: Technician
Experience: 1-3 Years
Qualification: Diploma/ITI
Key skill or job description: Handling dispensing of Raw Material & Packing Material and Finished Goods.
11. Department: Quality Control (OSD)
Designation: Officer to Sr. Executive
Experience: 4-7 Years
Qualification: M.Sc./ M. Pharma/ B. Pharma
Key skill or job description: Well versed with Analytical methods validation/Method transfer/Cleaning validation, Handling HPLC, GC, DR and other instruments, Reviewing analytical data. Preparation of Validation protocols and reports. QC analyzing, Stability, In process, FP, RM sample. Handling instruments (HPLC, GC, FTIR, UV, DR, SSA), and review of analytical data.
12. Department: Production (OSD)
Designation: Officer to Executive
Experience: 2-7 Years
Qualification: M. Pharma/ B. Pharma
Key skill or job description: Granulation / Compression / Packing. Audit exposure in USFDA/EUGMP approved company. Exposure of Material Handling / Electronic Batch Record filling and review / Good Knowledge of SAP System & Their T Codes for PP_PI / Machine Alarms Handling / QMS / Deviation / Change Control / SOP / Daily Reports Preparation/ Manpower Handling.
13. Department: Production (OSD)
Designation: Technician
Experience: 2-6 years
Qualification: D. Pharma/ Diploma/ ITI
Key skill or job description: Granulation / Compression / Liquid & Paste Manufacturing / Liquid Filling / Bottle Primary Packing. Audit exposure in USFDA/EUGMP approved company. Independently handle the granulation area equipment. Expertise in Top spraying and roll compaction process. Handle the Liquid and Paste Manufacturing equipment’s and their process. Working in Primary Bottle Packing (CVC Line) / Liquid Filling. Good Computer knowledge / Machine Operation / Cleaning / Changeover / Alarm Handling.
14. Department: Projects
Designation: Assistant Manager
Experience: 8-10 Years
Qualification: B.E / B. Tech – Instrumentation/ Electronics
Key skill or job description: Exposure of execute and manage instrumentation works. Exposure of design, execution, and maintenance of instrumentation and control systems, with strong hands-on exposure to BMS, EMS, and DCS in industrial or infrastructure projects. Experience in installation, commissioning, and system integration.
15. Department: Projects
Designation: Assistant Manager
Experience: 8-10 Years
Qualification: B.E / B. Tech – Electrical
Key skill or job description: Experience of design and execution on of projects, should have basic knowledge of utility and facility design, should have experience of working in projects, experience of project and construction management.
16. Department: Projects
Designation: Officer
Experience: 5-6 Years
Qualification: B.E / B. Tech – Mechanical
Key skill or job description: Knowledge of Execution, Installation, and Maintenance of mechanical systems, ensuring compliance with project specifications, safety norms, and quality standards. Ability to coordinate effectively with vendors and project teams.
Work Location: Pithampur, Madhya Pradesh
Date: 10th May 2026 (Sunday).
Time: 9.30 AM – 3.30 PM.
Venue: Hotel The Red Maple, Jhoomar Ghat, Rasalpura, Rau, Indore, Madhya Pradesh – 453446.
Candidates who cannot come for the interview can share resume on this mail id: [email protected]
Important Notes
* Candidates should have exposure with the USFDA approved company.
* Candidates should have good communication skills.
* Contact No. 9109996408