RPG Life Sciences-Walk in Interview for Production / Packing / QA / QC on 8th March 2026
RPG Life Sciences, a part of RPG Enterprises, is an integrated pharmaceutical company operating in the domestic and international markets in the Branded Formulations, Global Generics and Synthetic APIs space. The company is a research based, pharmaceutical company, producing a wide range of quality, affordable medicines.
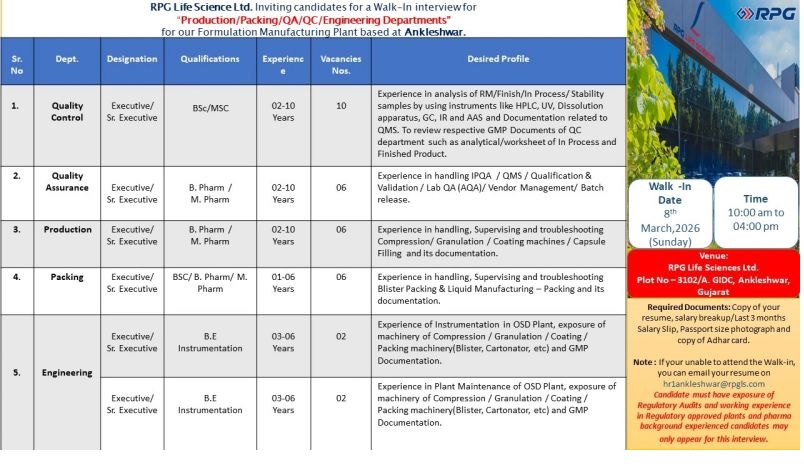
RPG Life Science Ltd. Inviting candidates for a Walk-In interview for “Production/Packing/QA/QC Departments” for our Formulation Manufacturing Plant based at Ankleshwar.
Quality Control
Post : Executive / Sr. Executive
Qualification : BSc/MSc
Experience : 02-10 years
No of posts : 10
Desired Profile : Experience in analysis of RM/Finish/ln Process/Stability samples by using instruments like HPLC, UV, Dissolution apparatus, GC, IR and AAS and Documentation related to QMS. To review respective GMP Documents of QC department such as analytical/worksheet of In Process and Finished Product.
Quality Assurance
Post : Executive / Sr. Executive
Qualification : B.Pharm, M.Pharm
Experience : 02-10 years
No of posts : 06
Desired Profile : Experience in handling IPQA / QMS / Qualification & Validation / Lab QA (AQA)/Vendor Management/ Batch release.
Production
Post : Executive / Sr. Executive
Qualification : B.Pharm, M.Pharm
Experience : 02-10 years
No of posts : 06
Desired Profile : Experience in handling, Supervising and troubleshooting Compression/ Granulation / Coating machines / Capsule Filling and its documentation.
Packing
Post : Executive / Sr. Executive
Qualification : BSc, B.Pharm, M.Pharm
Experience : 01-06 years
No of posts : 06
Desired Profile : Experience in handling, Supervising and troubleshooting Blister Packing & Liquid Manufacturing – Packing and its documentation
Walk in Interview
Date : 8th March 2026
Time : 10:00 am to 04:00 pm
Venue : RPG Life Sciences Ltd. Plot No-3102/A. GIDC, Ankleshwar, Gujarat
Required Documents : Copy of your resume, salary breakup/Last 3 months Salary Slip, Passport size photograph and copy of Adhar card. Note: If your unable to attend the Walk-in, you can email your resume on [email protected]
Candidate must have exposure of Regulatory Audits and working experience in Regulatory approved plants and pharma background experienced candidates may only appear for this interview.