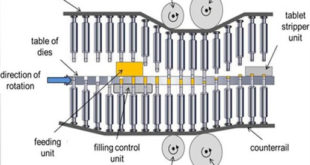
TABLET TOOLING Basics of Tablet Tooling Tablet compression machines are made keeping in view the type of dies and punches will be used on them. The dies and punches and their setup on compression machine is called tooling, it is classified as B and D mainly. The B tooling dies …
Read More »Preformulation for Tablets, Capsules, Liquid Orals
Preformulation for Tablets, Capsules, Liquid Orals Before developing a formulation like tablets, capsules, liquid orals we study the suitability of new drug or drug and excipients for the chosen formulation which is called preformulation. Preformulation Definition Preformulation may be defined as a stage of the research and development process where …
Read More »MONOGRAPH ON LIQUID PREPARATIONS FOR ORAL USE AS PER INTERNATIONAL PHARMACOPOEIA
MONOGRAPH ON LIQUID PREPARATIONS FOR ORAL USE AS PER INTERNATIONAL PHARMACOPOEIA Definition Liquid preparations for oral use are usually solutions, emulsions or suspensions containing one or more active ingredients in a suitable vehicle, they may in some cases consist simply of a liquid active ingredient used as such. Liquid preparations …
Read More »Aceclofenac 100 mg Film-coated Tablets
Aceclofenac 100 mg Film-coated Tablets 1. Name of the medicinal product Aceclofenac 100 mg Film-coated Tablets 2. Qualitative and quantitative composition Each film-coated tablet contains 100 mg of aceclofenac For the full list of excipients, see section 6.1. 3. Pharmaceutical form Film-coated tablet. White, round, biconvex film-coated tablets, 8 mm …
Read More »Actelsar HCT tablets
Actelsar HCT tablets 1. Name of the medicinal product Actelsar HCT 40 mg/12.5 mg tablets Actelsar HCT 80 mg/12.5 mg tablets 2. Qualitative and quantitative composition Actelsar HCT 40 mg/12.5 mg tablets Each tablet contains 40 mg telmisartan and 12.5 mg hydrochlorothiazide. Actelsar HCT 80 mg/12.5 mg tablets Each tablet …
Read More »Actos tablets
Actos tablets 1. Name of the medicinal product Actos 15 mg tablets Actos 30 mg tablets Actos 45 mg tablets 2. Qualitative and quantitative composition Actos 15 mg tablets Each tablet contains 15 mg of pioglitazone (as hydrochloride). Excipient with known effect Each tablet contains 92.87 mg of lactose monohydrate …
Read More »ABILIFY 1 mg/mL oral solution
ABILIFY 1 mg/mL oral solution 1. Name of the medicinal product ABILIFY 1 mg/mL oral solution 2. Qualitative and quantitative composition Each mL oral solution contains 1 mg of aripiprazole. Excipients with known effect (per mL) 200 mg fructose, 400 mg sucrose, 1.8 mg methyl parahydroxybenzoate (E218), 0.2 mg propyl …
Read More »Pharmaceutical Development as per USFDA
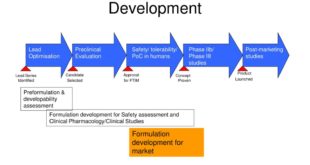
PHARMACEUTICAL DEVELOPMENT I. INTRODUCTION II. PHARMACEUTICAL DEVELOPMENT A. Components of the Drug Product 1. Drug Substance 2. Excipients B. Drug Product 1. Formulation Development 2. Overages 3. Physicochemical and Biological Properties C. Manufacturing Process Development D. Container Closure System E. Microbiological Attributes F. Compatibility III. GLOSSARY INTRODUCTION The Pharmaceutical …
Read More »INPROCESS CONTROL OF CAPSULE MANUFACTURING
INPROCESS CONTROL OF CAPSULE MANUFACTURING OBJECTIVE: To lay down the procedure for In process Control of Capsule Manufacturing. SCOPE: This SOP covers the responsibility and procedure for In process control during capsule manufacturing. RESPONSIBILITY: In process Quality Assurance Executive/Officer. ACCOUNTABILITY: Quality Assurance Manager. PROCEDURE: Carry out line clearance at each …
Read More »SOP For MFR For Ornidazole Tablets 500 mg
SOP For MFR For Ornidazole Tablets 500 mg Label Сlaim Manufacturing Formula List of Equipment Manufacturing Instructions Manufacturing Process Flow Chart Manufacturing Process Details Packaging Finished Product Specifications Procedural Changes Label Сlaim Product Ornidazole Tablets 500 mg Strength 500 mg Label сlaim Each Film uncoated tablet contains: Ornidazole …………500 …
Read More »MASTER MANUFACTURING FORMULA OF LISINOPRIL & HYDROCHLOROTHIAZIDE 10+12.5 MG TABLETS
MASTER MANUFACTURING FORMULA OF LISINOPRIL & HYDROCHLOROTHIAZIDE 10+12.5 MG TABLETS Contains Label Сlaim Manufacturing Formula List of Equipment Manufacturing Instructions Manufacturing Process Flow Chart Manufacturing Process Details Packaging Finished Product Specifications Procedural Changes Label Сlaim Product Lisinopril & Hydrochlorothiazide Tablets Strength 10+12.5 mg Label сlaim Each tablet contains: Lisinopril …
Read More »SOP for sterilization of Miscellaneous Items
SOP for sterilization of Miscellaneous Items Objective: To lay down a procedure for sterilization of miscellaneous Items. Scope: This SOP is applicable for sterilization of Miscellaneous Items. Responsibility: Officers/above (Production-Injectable) is responsible for sterilization of Miscellaneous Items. Accountability: Production Head is accountable for the implementation and compliance of this SOP. …
Read More »SOP for sterilization of cartridge Filter
SOP for sterilization of cartridge Filter Objective: To lay down a procedure for sterilization of cartridge filter. Scope: This SOP is applicable for sterilization of cartridge filter pre assembled in filter housings. Responsibility: Production Officers / Executive. Accountability: Production Head shall be accountable for the compliance of SOP. Abbreviations SOP …
Read More »Cartridge Filter
What is a Cartridge Filter A cartridge filter is a type of filter that employs a porous cartridge as the medium for trapping impurities from liquids or gases. It is usually made of materials like paper, polyester, or other synthetic fibers, and its design allows for efficient filtration by trapping …
Read More »SOP for delabelling and repacking of vials
SOP for delabelling and repacking of vials Objective: To lay down a procedure for Delabelling and Repacking of vials. Scope This SOP is applicable on procedure for Delabelling and Repacking of vials for Production dry powder Injection facility. Responsibility Production Officers / Executive. Accountability Production Head shall be accountable for …
Read More »SOP for cleaning of vial filling machine and its parts
SOP for cleaning of vial filling machine and its parts Objective To lay down a procedure for cleaning of vial filling machine and its parts. Scope This SOP is applicable for cleaning of vial filling machine and its parts for Production dry powder Injection facility. Responsibility Production Officers / Executive. …
Read More »SOP on cleaning of Liquid filling machine (Watson Marlon )
SOP on cleaning of Liquid filling machine (Watson Marlon ) Objective: To lay down the procedure for cleaning of liquid filling machine (Watson Marlon). Scope This SOP is applicable for the procedure of cleaning of liquid filling machine shall be accountable for the compliance of SOP. Responsibility Production Officers / …
Read More »SOP for operation of sticker labeling machine (Maharshi)
SOP for operation of sticker labeling machine (Maharshi) Objective: To lay down a procedure for operation of sticker labeling machine. Scope: This SOP is applicable for sticker labeling machine. Responsibility: Production operator/Technician- responsible for operation of sticker labeling machine. Production officer/ Executive – to ensure that operation of sticker labeling …
Read More »SOP for operation of shrink wrapping machine
SOP for operation of shrink wrapping machine Objective: To lay down a procedure for operation of shrink wrapping machine. Scope: This SOP is applicable for shrink wrapping machine. Responsibility: Officers or above – Production. Accountability: Production Head. Abbreviations SOP : Standard Operating Procedure. MASTER FORMULA OF MEBENDAZOLE TABLETS Procedure: Pre-startup: …
Read More »SOP on cleaning of shrink wrapping machine
SOP on cleaning of shrink wrapping machine Objective: To lay down a procedure for cleaning of shrink wrapping machine. Scope This SOP is applicable for cleaning of shrink wrapping machine in production area. Responsibility Production operator / technician – responsible for cleaning of machine. Production officer / Executive – to …
Read More »